|
3/22/2021 0 Comments Compound Calculator Chemistry
From there, you calculate the ratios of different types of atoms in the compound.For example, if you assume that you have 100 g of a compound composed of 60.3 magnesium and 39.7 oxygen, you know that you have 60.3 g of magnesium and 39.7 g of oxygen.The only time you dont do this is if the problem specifically gives you the masses of each element present in the unknown compound.).
Then divide all the mole values you calculated in Step 2 by this smallest value. 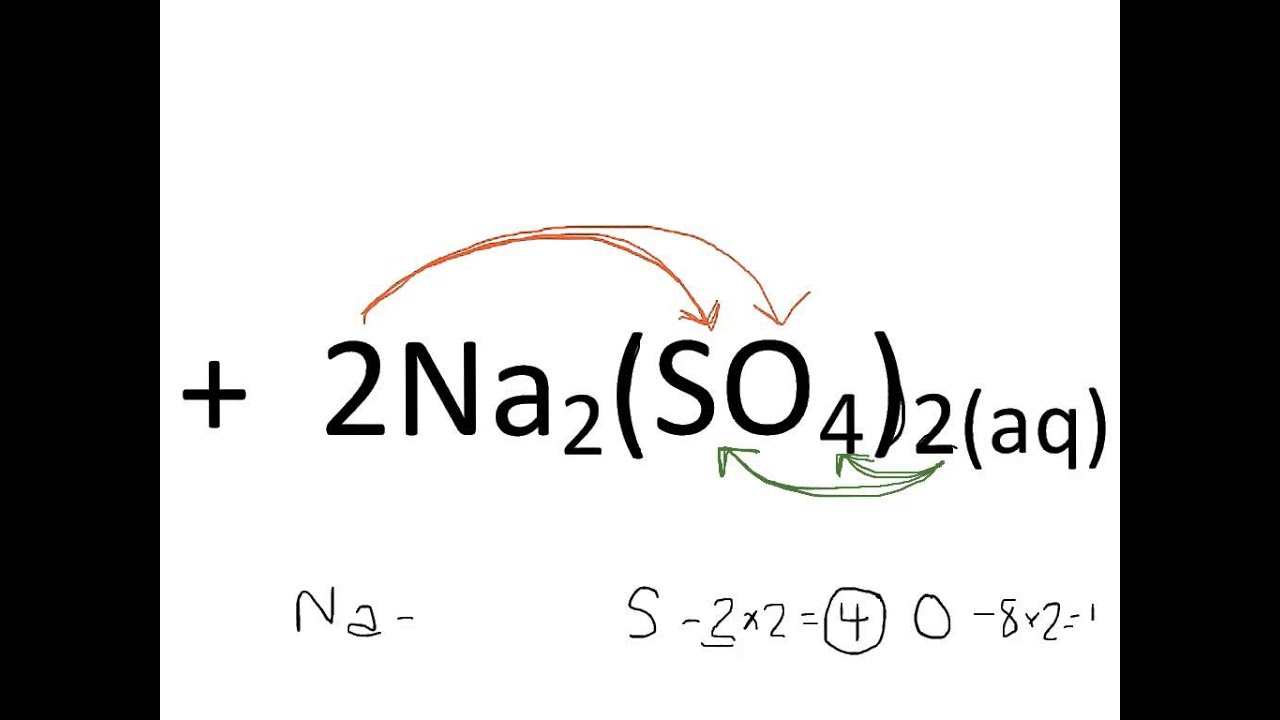
Far more likely is that the atoms of nitrogen and oxygen are combining in a 1: 0.5 ratio but do so in a larger but equivalent ratio of 2: 1. 
Therefore, you have 40.0 g of carbon, 6.7 g of hydrogen, and 53.3 g of oxygen. Convert each of these masses to moles by using the gram atomic masses of C, H, and O. Next, divide all the mole numbers by the smallest among them, which is 3.33. This division yields. The actual number of atoms within each particle of the compound is some multiple of the numbers expressed in this formula.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
